SiO2 Materials Sciences launches a line of specialty vials for biological drugs and vaccines. The SiO2 vials solve significant challenges in the commercialization of vaccines and biological drugs which presently cannot be solved by glass or plastic vials.
According to Chief Scientist Chris Weikart, “Many drug development and drug formulation innovations can be limited due to variables associated with traditional glass vials (lot to lot material variability, leachables, etc.). These variables result in lost time and money due to extensive formulation studies. With the SiO2 Vials, we eliminate these variables and allow our drug development partners to bring their innovations to life.”
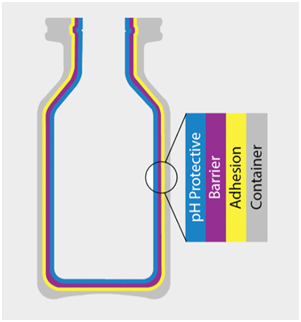
SiO2 Vials:
Thermal Stability & Integrity: with a thermal stability range of -196˚C to 121˚C and with a seal integrity of -80˚C to 25˚C, the SiO2 vials provide the ideal primary container for therapies which require cold storage
Chemical Stability: safe and suitable for a wide range of drug characterizations including pH ranges from 3-12; there is no risk of delamination as observed with glass vials
Gas barrier: SiO2 vials have gas permeation properties like glass and therefore provide the required shelf life and stability required by many therapies
Mechanical durability: SiO2 vials can withstand 1,500 pounds of direct force saving millions from breaking on filling lines and minimizing loss in the supply chain
No Breakage: the vials are shatterproof making them safe for healthcare staff and the patient
SiO2’s Science:
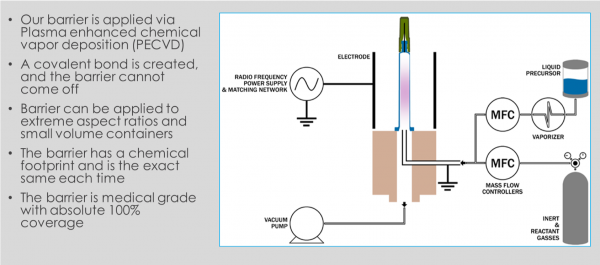
“While the vials themselves share many properties of both glass and plastic, we have conducted and passed all the studies required for regulators to approve the SiO2 Vials as a like for like to glass,” stated Pete Sagona, Senior Vice President.
According to Lawrence Ganti, Chief Business Officer, “SiO2 is dramatically ramping up capacity to meet customer demands and to ensure that the surge seen due to the COVID19 vaccine developments can be accommodated. We have very short lead times, so we can accommodate the surge in demand for glass or plastic vials.”
The company launched the vials commercially on April 22, 2020 and are ready to partner with companies to fast-track their treatments and vaccines.
To know more, visit www.sio2ms.com or email, info@sio2ms.com.
Media Contact
Company Name: SiO2 Material Science
Contact Person: John Kim
Email: Send Email
Phone: 8449309323
City: Auburn
State: Alabama
Country: United States
Website: http://sio2ms.com